Process of reassessing guaranteed healthcare services
The process of reassessing guaranteed healthcare services in a given clinical area, in the context of a review of the manner in which health problems are diagnosed and treated, including changes in the conditions of implementation or financing of those services, requires a complex methodological approach and, consequently, its implementation period is long-term. It results from the multi-stage and complex analytical processes, including the simultaneous involvement of several analytical teams and clinical experts.
The aim of the actions concerning proposals for changes in the area of the health benefit package is to provide the necessary healthcare services of proven efficacy which are most important from the point of view of the society’s health, which can be offered to beneficiaries within the framework of the available common insurance funds.
These actions take into account the perspective of different stakeholders in the healthcare system:
- patients,
- healthcare providers,
- the public payer,
- the Minister of Health.
The goal of the reassessment of the healthcare benefit package include:
- simplification of the current provisions by changing / unifying the form of description of guaranteed healthcare services;
- clarification of the criteria for the eligibility of patients for particular healthcare services;
- (as a rule) presentation of the list of guaranteed healthcare services through health technologies (procedures in clinical indication) – some of the procedures will remain covered in their full scope of application when backed by the available evidence;
- verification of the validity of the current quality requirements of individual healthcare services (conditions of implementation);
- identification of optimal conditions for the provision of healthcare services (organisational models), ensuring the highest efficacy and cost effectiveness of guaranteed services;
- reduction of expenditure on healthcare services with proven inefficiency.
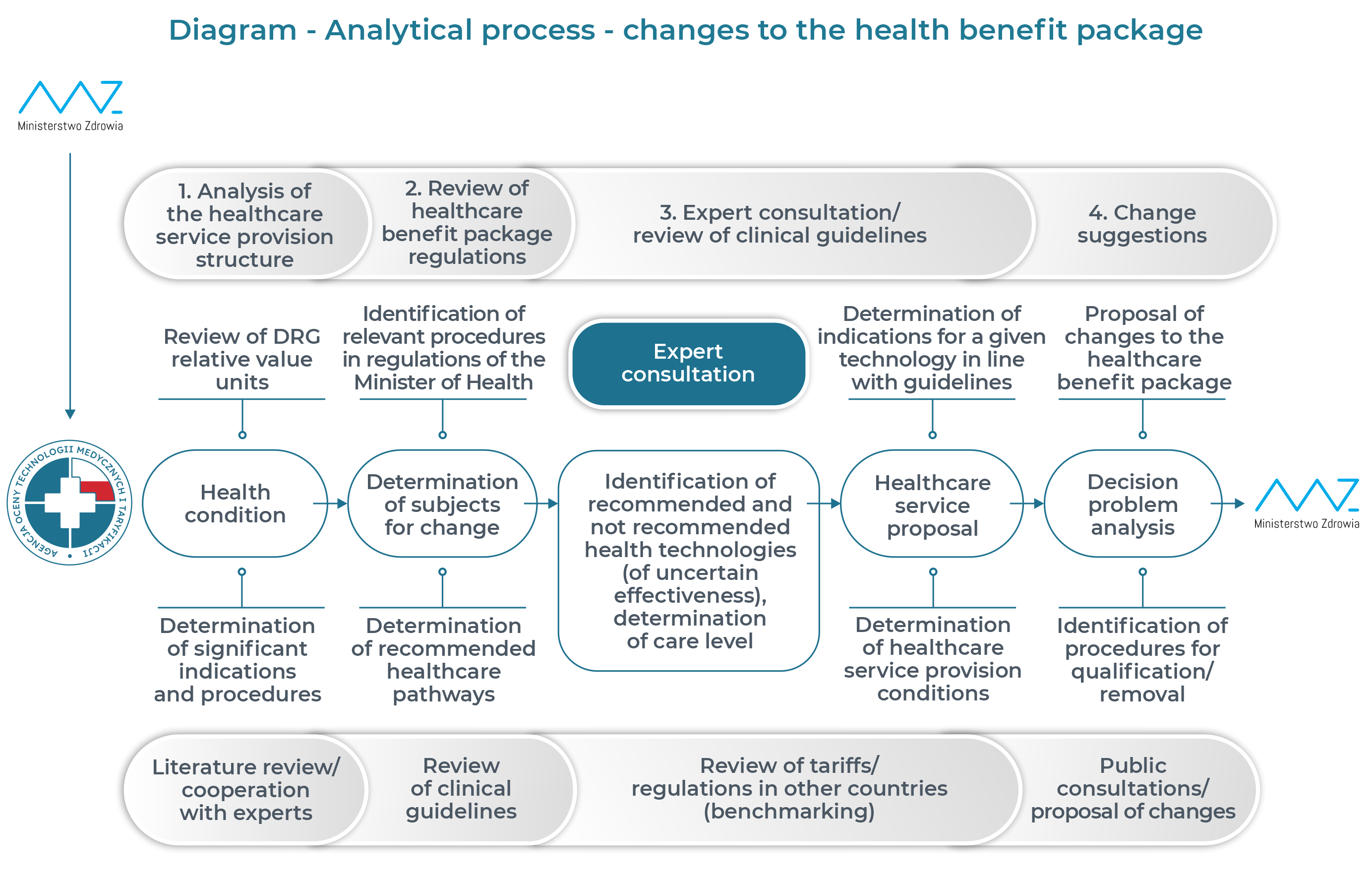
Main directions of the analytical process of evaluating guaranteed healthcare services:
- problem identification – analysis of health needs, the necessity of providing service in the context of the clinical condition, etc.;
- problem selection – identification of priority areas requiring implementation of changes, including the analysis of changes in a given area suggested by experts;
- review of the healthcare services currently included in the health benefit package in the scope of primary healthcare, outpatient specialist care, hospital treatment – analysis of the conditions for their implementation and settlement;
- review and verification of current clinical guidelines in Poland and worldwide in terms of recommended diagnostic and therapeutic practice in a given medical indication together with assessment of their quality – AGREE II;
- verification of medical procedures currently included in the health benefit package (primary healthcare, outpatient specialist care, hospital treatment) in relation to the recommended diagnostic and therapeutic procedures, aimed at specifying the following procedures:
– recommended and not included in the package but necessary in the context of the clinical condition,
– not recommended (identification of procedures with doubtful/uncertain efficacy or uncertain safety profile);
- consultation process with the clinical experts and the National Health Fund;
- determination of the directions of necessary changes in the health benefit package.
*Ladies and Gentlemen
We inform you that the materials and results of the discussions posted on the Agency’s website are the result of the conceptual work and analytical process carried out by the team of the Agency for Health Technology Assessment and Tariff System based on the EBM paradigm, including: search, selection, synthesis and interpretation of scientific evidence, or the data analysis carried out.
In connection with the above, we would like to inform you that the use of analytical material or the results of the discussion, in accordance with good practice, should be accompanied by information on the source in the form: [title of presentation / report], AOTMiT, Warsaw, June 2021]

