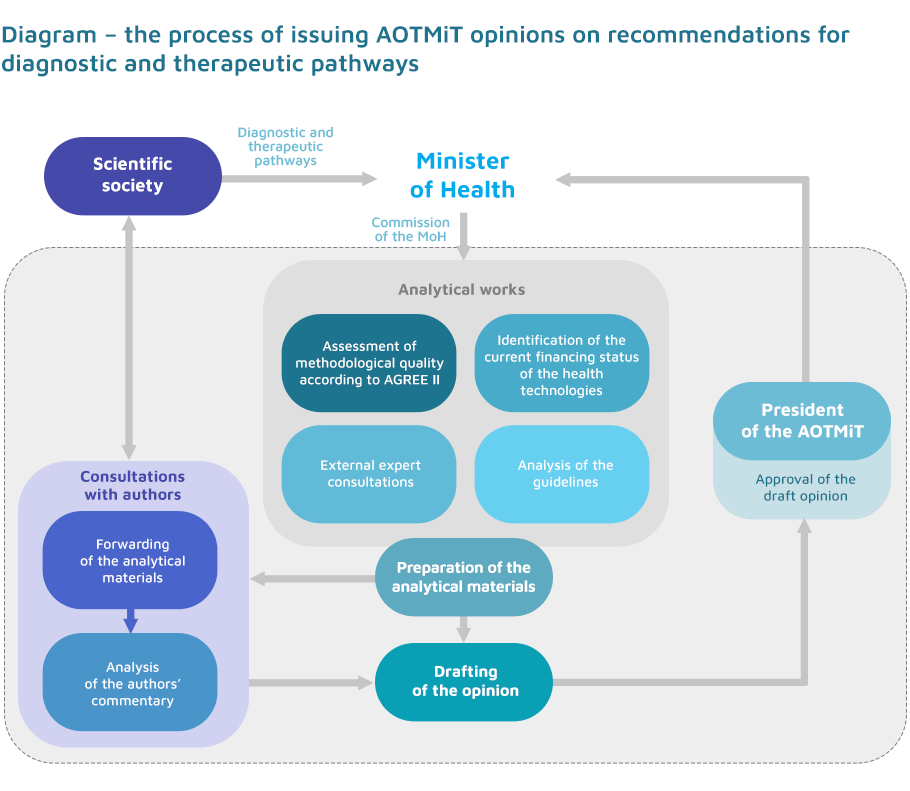
The Minister of Health commissions the Agency for Health Technology Assessment and Tariff System to assess recommendations on diagnostic and therapeutic pathways before publication.
The main purpose of the process is to identify recommendations that could be implemented in the healthcare system in order to improve the quality of the treatment process.
The analytical works undertaken in the Agency are carried out according to strictly defined procedures.
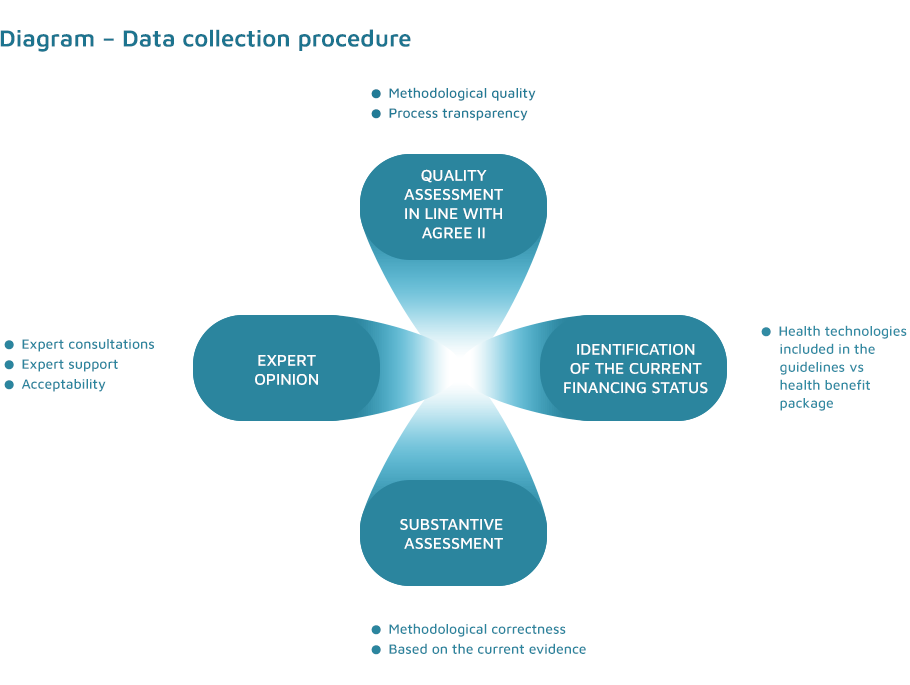
The procedures aim at objectively compiling the data collected in four areas:
After the analytical works are completed, the document prepared during the substantive assessment is consulted with the authors of the recommendations, who also address the Agency’s remarks..
*Ladies and Gentlemen
We inform you that the materials and results of the discussions posted on the Agency’s website are the result of the conceptual work and analytical process carried out by the team of the Agency for Health Technology Assessment and Tariff System based on the EBM paradigm, including: search, selection, synthesis and interpretation of scientific evidence, or the data analysis carried out.
In connection with the above, we would like to inform you that the use of analytical material or the results of the discussion, in accordance with good practice, should be accompanied by information on the source in the form: [title of presentation / report], AOTMiT, Warsaw, June 2021]

