“Our mission is to make the healthcare system more efficient. We use EBM and HTA to facilitate the decision-making process with regard to the financing of healthcare services. Our analyses and assessments are comprehensive. We ensure the highest quality of all our activities”.
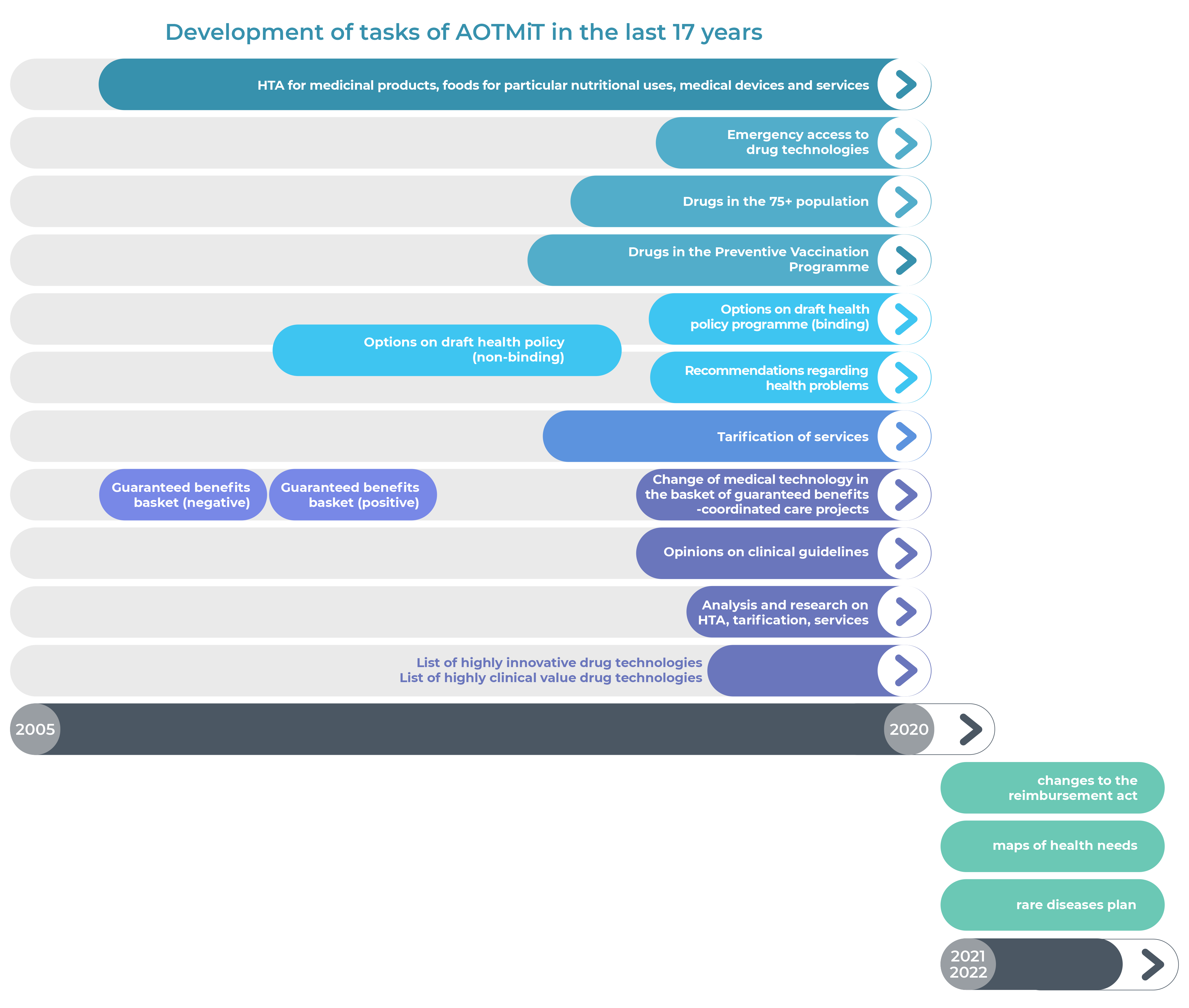
The Agency was created in 2005; since then we have become one of the strongest and most recognised European institutions in the field of assessment and valuation of healthcare services. The Agency intends to maintain its leading position in the CEE region.
Pursuant to the Act of 25 June 2009 amending the Act on healthcare services financed from public funds and the Act on prices, we have obtained the status of a public entity with legal personality.
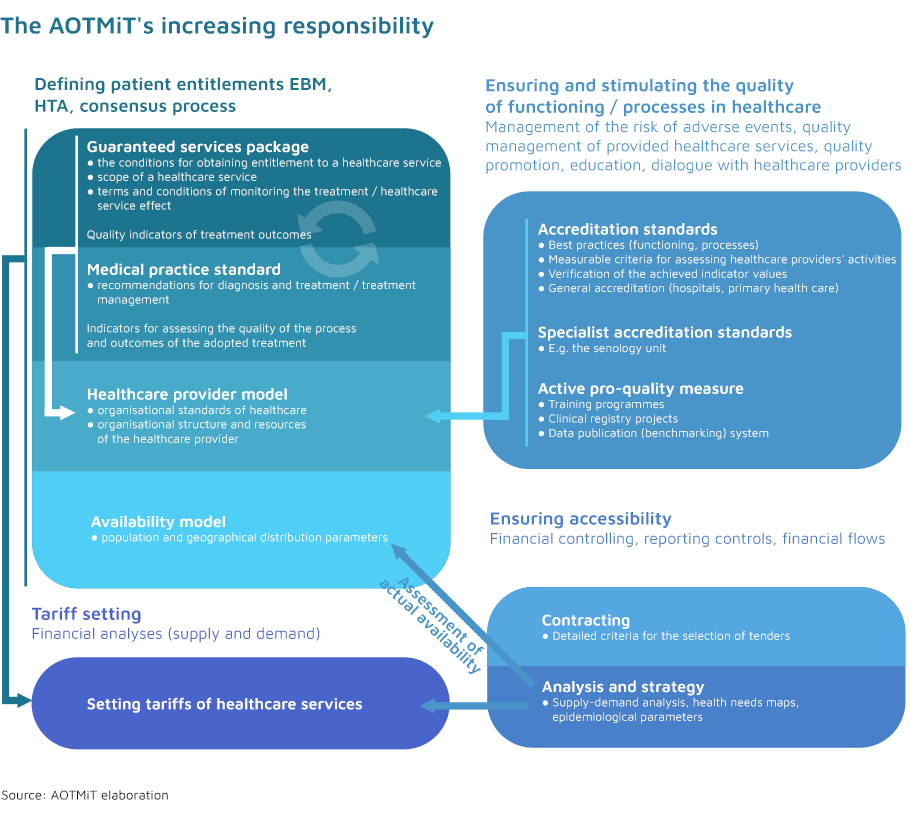
- As a public HTA institution, we support the decision-making process in the healthcare system (in a transparent process) by providing comprehensive health technology assessment information, which ensures access to the system and its cost-effectiveness.
- We are involved in both scientific research and development activities in Poland and abroad.
- Our priorities stem from the needs of the Polish healthcare system and our practices are based on documented methods and guidelines reflecting the latest approaches to evidence-based decision making.
- It is our intention to ensure that we always remain a responsible partner by building trust-based relations as well as focusing on dialogue and transparent cooperation.
Over the years, both the role and the tasks of the Agency have changed significantly. Taking up new challenges and implementing the entrusted tasks enables continuous development and strengthens the Agency’s international position
*Ladies and Gentlemen
We inform you that the materials and results of the discussions posted on the Agency’s website are the result of the conceptual work and analytical process carried out by the team of the Agency for Health Technology Assessment and Tariff System based on the EBM paradigm, including: search, selection, synthesis and interpretation of scientific evidence, or the data analysis carried out.
In connection with the above, we would like to inform you that the use of analytical material or the results of the discussion, in accordance with good practice, should be accompanied by information on the source in the form: [title of presentation / report], AOTMiT, Warsaw, June 2021]